What are our clients doing to help stop COVID-19?
Written by:
Azeem Zeekrya
Date:
COVID-19 is not an ordinary disease. It’s highly contagious and much more fatal than the usual respiratory disease — two key differentiators that have caused the world to come to a halt.
As ordinary citizens, like HDMZers, do their part to help curb its spread, doctors and nurses are tending to patients on the front lines and life sciences organizations are currently conducting more than 200 clinical trials to develop the treatments and vaccines for COVID-19. This includes many of HDMZ’s clients, who are doing what they do best: using science to solve this problem.
Knowledge is Power
Diagnostic testing is essential during a pandemic, helping medical professionals make informed public health decisions about the scale of the outbreak, the speed of its spread and its lethality.
A common method for testing for viruses is RT-PCR, which reverses and amplifies the target viral RNA in a patient’s sample back to DNA for detection, if it’s present. Both ThermoFisher Scientific and Cepheid have developed RT-PCR tests that have been given emergency authorization by the FDA to diagnose patients. Cepheid’s test is particularly interesting because the entire RT-PCR reactions are contained inside a small plastic cartridge.

Cepheid’s Xpert Xpress SARS-CoV-2 houses the full RT-PCR process in its cartridge.
With earlier testing shortfalls, many people who had caught COVID-19 may have gone unnoticed. While RT-PCR tests help identify active cases, antibody assays detect whether a person has already recovered and developed immunity. Beckman Coulter is developing an assay that looks for anti-SARS-CoV-2 antibodies in a person’s blood to determine just that.
Boosting Immunity
Vaccines help build herd immunity and will be the essential safeguard for preventing a devastating second round of COVID-19 infections. In a show of partnership, GSK is making its vaccine adjuvants available for other groups to make use of. Adjuvants increase the effectiveness of a vaccine by stimulating the immune system with less of the viral antigen. This allows more doses of a vaccine to be produced and can also lift some of the supply concerns around vaccine production for a disease as widespread as COVID-19.
Attacking the RNA
At their core, viruses are genomic hijackers, and SARS-CoV-2 (which causes COVID-19) is no exception. Genetic tools are a necessity for combatting them and this is right up Twist Bioscience’s alley. It used its know-how to create synthetic copies of the virus’ RNA so that scientists can work without exposing themselves to the active virus. Twist is also producing positive RNA controls for COVID-19, which help validate whether diagnostics are giving accurate results.
RNA is also a drug target and Gilead’s remdesivir is one such candidate. This antiviral works against a wide range of viruses, possibly including SARS-CoV-2, by disrupting RNA replication, so that when a virus copies itself, its clones lack a full genome and lose the ability to infect other cells. Remdesivir is currently in clinical trials for COVID-19.
Antibodies offer another course of action outside of RNA targeting, such as treatments that use antibodies effective against SARS-CoV-2. Twist’s synthetic biology capabilities allow it to write the exact antibody sequence needed and it’s partnering with Vanderbilt University to isolate antibodies from patients who’ve recovered from COVID-19 to gather a library of possible sequences.
Science Must Go On
Even though it feels as if the world has stopped to deal with COVID-19, other diseases are still affecting millions of patients. With strict border lockdowns and reduced flights because of the disease, we have been plunged into a logistical nightmare.
Marken is confronting this reality head on as it moves critical treatments and biological samples across borders amidst this crisis, while meeting the necessary preservation demands. Marken is also helping patients access its life-saving medications, such as in the case of an Italian man stranded in Saudi Arabia and a Canadian family who lost their medicine supply after the US-Canada border was shut down.
Clinical trials for other diseases are still ongoing, although limited, and ICON is helping researchers stay on track through this disruption. ICON has also created an online Coronavirus dashboard that tracks clinical trials for COVID-19, its spread throughout the world and major news stories regarding it.
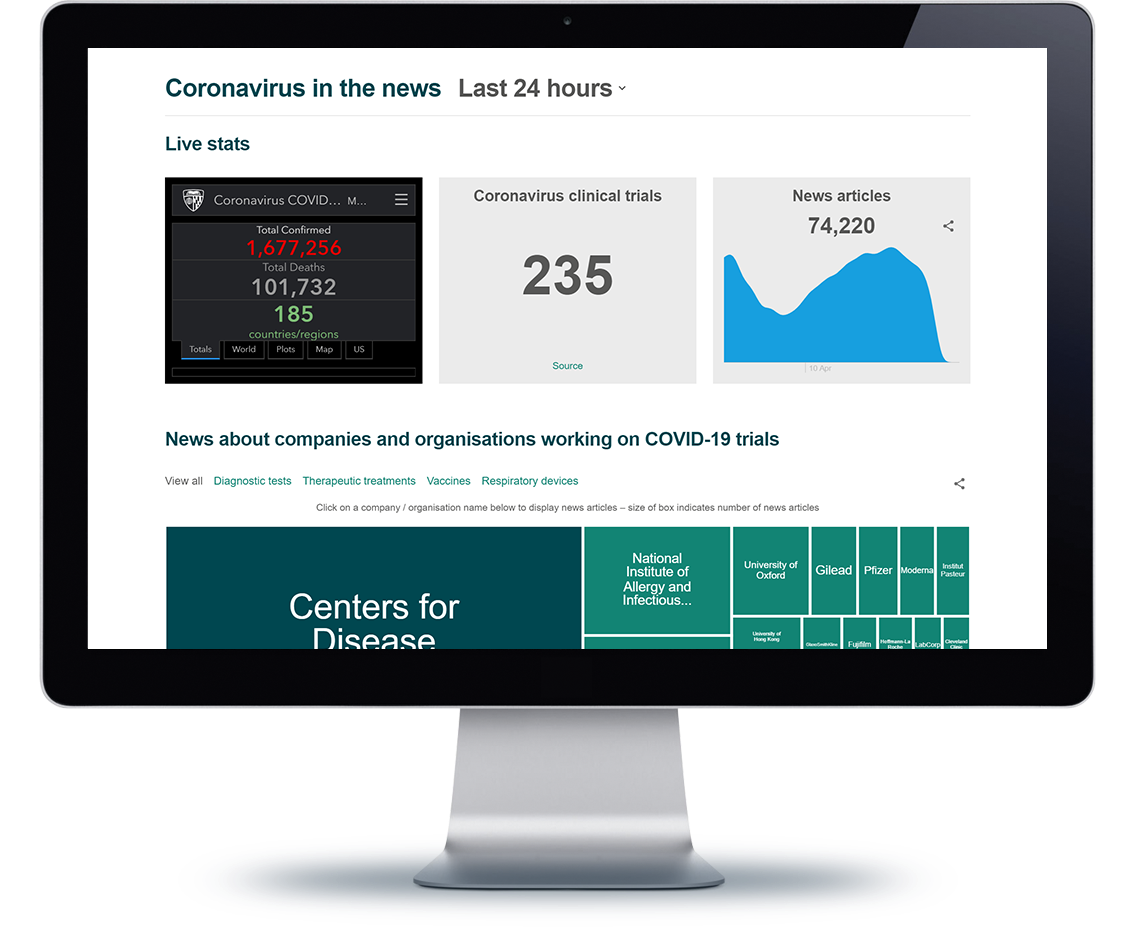
ICON’s Coronavirus Observatory gives a look into the latest figures, news and clinical trials for COVID-19
At HDMZ we believe that “science solves” and our clients are demonstrating that every day. We’re proud of serving those who are making the world a better place and of our role in helping them get the credit they deserve for doing so.
Written by

Azeem Zeekrya
Director, Scientific Communications
You may also like
All rights reserved Privacy Policy






